Huntington's Disease
MISBA® platform unlocks important targets with disease-modifying potential
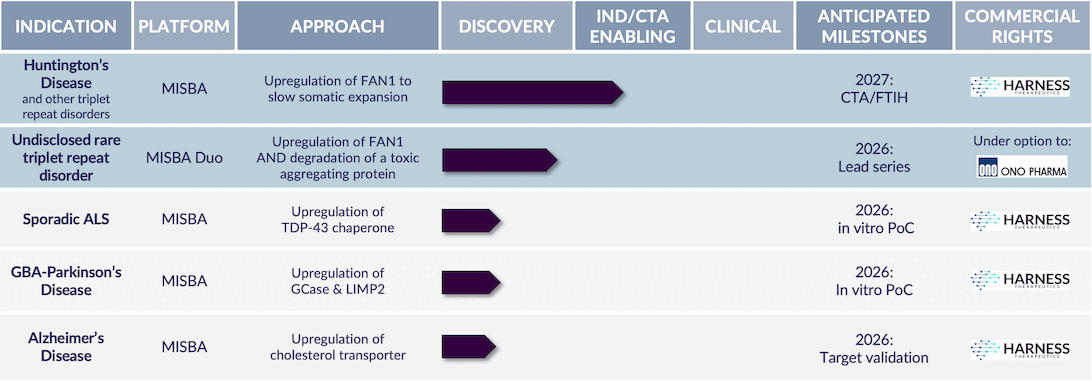
Progressing first-in-class validated targets for a range of large neurodegenerative disorders
Huntington’s disease: A fatal disorder with no effective treatment
Huntington’s disease (HD) is a devastating, inherited neurodegenerative disorder that causes progressive, irreversible brain deterioration. Symptoms typically emerge between age 30 and 50 and include movement disorders, cognitive decline, and psychiatric symptoms. There is currently no approved disease-modifying treatment, and affected individuals typically die within 15 years of symptom onset. An estimated 10–12 per 100,000 people are affected, with a similar number at risk or asymptomatic carriers.
Harness’s controlled, FAN1 upregulation approach to HD
The HD field has seen a shift over time regarding therapeutic strategies. Whilst historically, focus has been on reducing mutant Huntingtin protein (mHTT), these efforts have faced challenges with both safety and efficacy. Emerging evidence suggests that targeting mHTT alone is insufficient to halt disease progression.
More recent findings have demonstrated that slowing or halting the underlying process of the progressive somatic expansion of CAG trinucleotide repeats in the HTT gene offers the most promising opportunity to delay disease onset and progression.
Harness’ lead programme directly targets CAG repeat expansion by focussing on FAN1, a DNA repair nuclease with the strongest genetic association to disease onset. It has the potential to be a first and best-in-class disease-modifying therapy. The choice of FAN1 as a target is underpinned by the following characteristics:
- FAN1 is a key suppressor of somatic expansion, shown to slow CAG repeat growth in multiple independent studies
- Genome-wide association studies (GWAS) and SNP data demonstrate that natural variation in FAN1 levels correlates with delayed disease onset
- Experimental validation in patient-derived neuronal models shows that modest, controlled increases in FAN1 can safely and effectively slow the expansion process
HD therapeutic solution
Harness is developing a first-in-class, ASO-based therapy designed to precisely and safely upregulate FAN1 at the post-transcriptional level. Built on our proprietary MISBA®platform, the ASO construct:
- Provides self-limiting, controlled increases in FAN1 levels within a safe and efficacious range to avoid the risks associated with over-expression
- Is delivered directly into the CNS using established surgical techniques, reducing systemic exposure and mitigating oncogenic risk
- Uses clinically validated ASO chemistries to reduce developability risks
Our preclinical studies show that this approach significantly slows somatic expansion in disease-relevant neuronal models.
Beyond Huntington’s
FAN1’s role in triplet repeat expansion suppression extends beyond HD. This opens the door to future applications in other expansion-driven diseases, including:
- Spinocerebellar ataxias
- Fragile X-associated disorders
- Myotonic dystrophy type 1 (DM1)
Please contact us for further information about our Huntington's disease programme.